The safety debate we never had
Highlights
- Nearly all children across the globe are infected with rotavirus in the first three years of their lives, and they are “often asymptomatic.” There’s no medical treatment other than simply keeping the child hydrated with proper electrolytes.
- Not surprisingly, natural infection protects against future infection, even if asymptomatic. If a child does get reinfected, sickness is less severe and less likely to happen again.
- Justification for the rotavirus vaccine came from high numbers of deaths in young children in poor countries. But the first country to license it was the United States, and the rest of the world didn’t start adopting it into their immunization programs for years.
- Prior to the release of the vaccine, CDC scientists published a 1996 paper where they estimated less than 40 deaths due to rotavirus yearly before the vaccine was introduced.
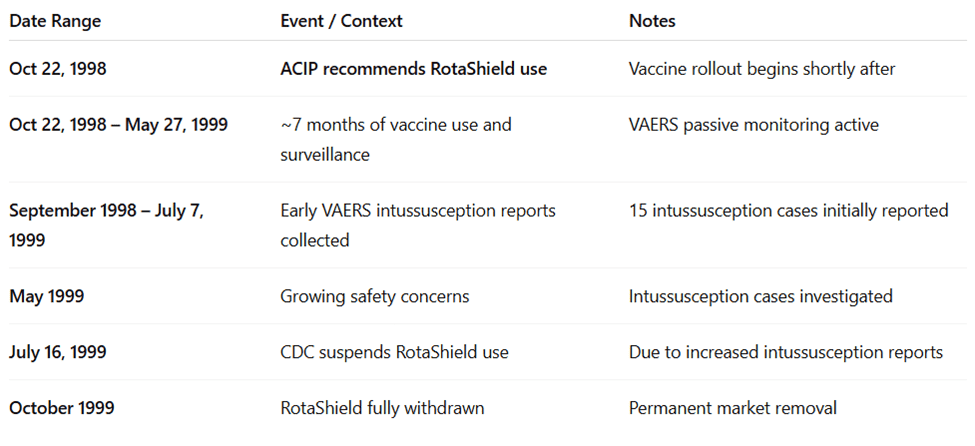
- RotaShield was licensed on August 31, 1998, and recommended by the CDC on October 22, 1998. Immediately, there were reports of intussusception (when the intestine folds in on itself) after vaccination.
- Rotavirus vaccines were initially removed from the childhood schedule due to VAERS reports revealing safety concerns, but were returned to the schedule after a few short years. And the new vaccines still cause the same injury as the original. We’re told it’s much less risky this time around, of course.
- After the ACIP deemed the vaccine unsafe for American children, it couldn’t be sold in other countries because it was publicly rejected. The ACIP even had discussions about how they would frame their announcement and withdraw so as to not affect the global market!
- Unfortunately, the rotavirus vaccine worked even less effectively in the very countries it was designed to help.“Rotavirus vaccines have also been shown to be 23% to 47% less effective in high child mortality regions compared to low child mortality regions.”
- Adding even more insult to injury here is the fact that the vaccine made the virus worse in the wild. Introduction of a vaccine into a field where the target can mutate creates pressure that drives strain replacement. In other words, the vaccine changes the illness, which, in turn, changes whether and how the vaccine will work.
- So does VAERS work? It sure does – even with very well-known flaws that lead to underreporting by magnitudes of 10s. What’s broken is the response to the safety signals.
- Compare intussusception numbers. They started investigating once nine reports were made, but look how many reports are in VAERS now! Why hasn’t the vaccine been pulled again?! What other vaccines carry reports that reflect a 1/10,000 risk of injury, or even 1/100,000?
- Medical ethics researcher Jason Schwartz put succinctly: The legacy of RotaShield was reduced to “little more than ‘the system worked so the system works.’” Parents are expected to believe that since one vaccine was publicly raked over the coals and temporarily removed from the constantly growing schedule, that they can sleep soundly, believing the government wouldn’t let a dangerous product be given to our kids.
Introduction
Rotavirus vaccine has the distinction of being the first and, for decades until COVID jabs were removed, the only vaccine taken off the CDC’s recommended childhood immunization schedule due to reports of injuries after being licensed and given to American children. (The diphtheria-pertussis-tetanus combination did change from a whole cell pertussis component to acellular after parents raised the alarm about vaccine injury in the 1970s and 1980s, but the combination was never removed from the schedule, nor was it discussed.)
Replacement rotavirus vaccines were returned to the schedule after a few short years; they were already in development as competitors when the first licensed vaccine was removed. Adding the vaccine back to the schedule was merely a matter of time, not a question of necessity. But it turns out the new vaccines still cause the same injury as the original. We’re told it’s much less risky this time around, of course.
How do the FDA and the CDC determine what level of risk is acceptable? What makes the rotavirus story unique? Read on to find some answers – and to discover more questions.
What is rotavirus
Nearly all children across the globe are infected with rotavirus in the first three years of their lives and they are “often asymptomatic.”1 Symptomatic rotavirus infections generally start with vomiting followed within two days by diarrhea.2 Most of these cases are mild but about a third of children get high fevers, according to the American Academy of Pediatrics’ “Red Book,” a pediatrician’s go-to reference for infectious disease. Illness lasts up to a week. “A small proportion (<1%) of children infected with rotavirus will develop dehydration” so severe they will need IV fluids and electrolytes, typically in the hospital.3 In these cases, it’s expected that a child will have fever and vomiting for three days and diarrhea for up to five days. The worst cases, and the most deaths, occur in developing countries where sanitation is poor, access to clean water is low, and malnourishment is rampant.
Transmission is believed to be fecal-oral, but may be respiratory, since there’s a seasonality similar to respiratory illnesses such as flu.4 The actual mode isn’t understood but the AAP claims rotavirus is hearty and can live “for weeks to months” in the environment, so could spread easily on things like toys.
There’s no medical treatment other than simply keeping the child hydrated with proper electrolytes. But protective and soothing effects have been found with breast milk and probiotics. A group of scientists and doctors who carefully study and summarize the best research on health and medicine around the globe, known as the Cochrane Review, looked at probiotic use in diarrheal disease and found it helpful along with rehydration to shorten the duration and severity of diarrhea.5 Further, the AAP Red Book lists breastfeeding as a “control measure,” telling physicians that “breastfeeding is associated with milder rotavirus disease and should be encouraged.”6 Shockingly, even Plotkin’s Vaccines (the go-to source for vaccine information) acknowledges, “Breastfeeding clearly protects against rotavirus disease.”
Infant diarrhea is not uncommon and there are many causes. How was rotavirus singled out for special treatment? It can’t be detected without lab testing,
In the 1970s, while newly emerging globalist giants were pushing plans to curb population growth (see Shining a light on tetanus),7 scientists wanted to know more about how long humans live, and why.8 Diarrhea topped the list of deadly illnesses for the youngest children around the globe, becoming a research topic for many scientists of the time.
Rotavirus was discovered in 1973 by Ruth Bishop. Bishop9 believed there was a bacteria to blame for fatal vomiting and diarrhea. She worked in her laboratory with samples from the Royal Children’s Hospital in Melbourne, Australia, for 12 years and found none. But when the electron microscope debuted, she set her sights on smaller culprits and what you seek you will find.10 What she and her colleagues saw with the new technology was described as a wheel-shaped virus. They hitched together the Latin words for wheel and poison to name it rotavirus.
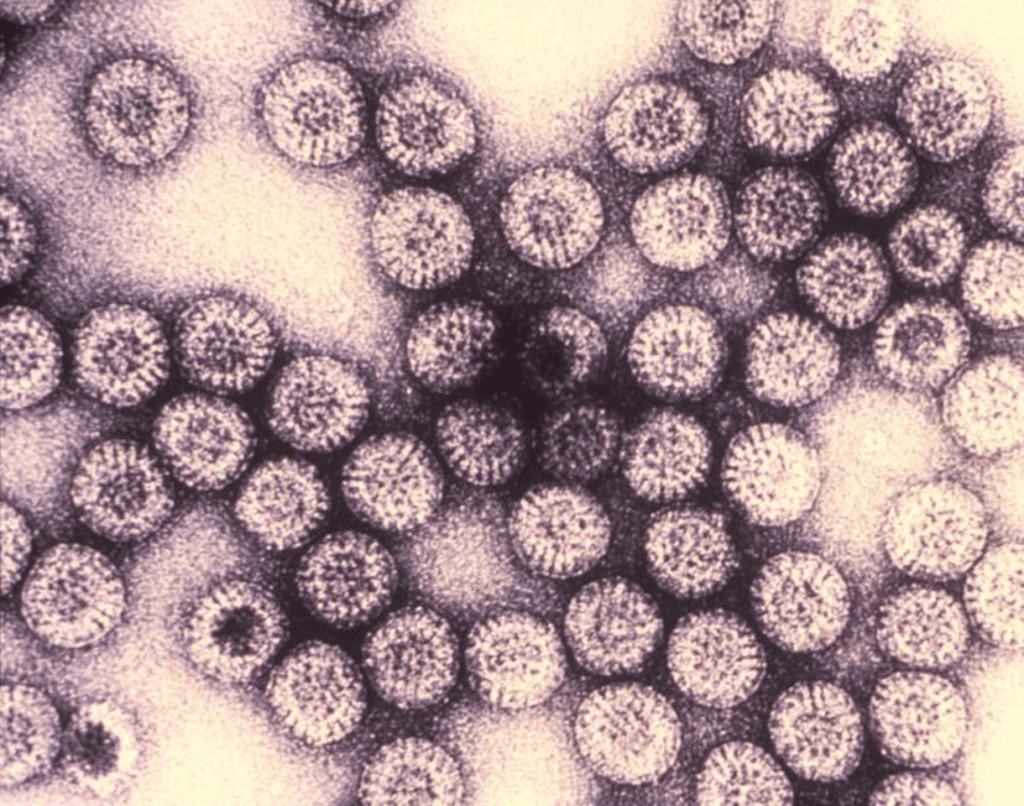
Despite being named after a flat object, rotavirus is actually a kind of sphere with 20 sides.11 It’s a classic virus structure with RNA inside a shell of layered proteins. The RNA is “segmented” which means it can break apart, rearrange, and create new strains with “mixed parentage.”12 This can happen naturally or be forced in a lab experiment. Because of this recombination, there are well over 100 strains,13 60 of which are known to cause illness in humans.14 But it is generally thought that only 5 strains cause 90% of known rotavirus illness in the U.S.15
Not surprisingly, natural infection protects against future infection, even if asymptomatic.16 If a child does get reinfected, sickness is less severe and less likely to happen again. A 1996 study in the New England Journal of Medicine (NEJM) concluded, “In infants, natural rotavirus infection confers protection against subsequent infection. This protection increases with each new infection and reduces the severity of the diarrhea.” In other words, if you get sick from one kind of rotavirus, your body learns to fight off that same kind pretty well, but you might still get a little sick from a different kind. Interestingly that paper is cited as laying the groundwork for the vaccine, because it proved natural immunity worked.17 Isn’t it interesting that proof of natural immunity is a foundation for vaccine science?

Who wanted a vaccine?
Justification for the rotavirus vaccine came from high numbers of deaths in young children in poor countries. But the first country to license it was the United States, and the rest of the world didn’t start adopting it into their immunization programs for years. In fact, developing countries initially rejected it. Why? Because the U.S. rolled it out first, took it off the schedule for safety reasons, but still attempted to convince developing countries to adopt it.
Prior to the release of the vaccine, CDC scientists published a paper in 1996 where they estimated less than 40 deaths due to rotavirus yearly before the vaccine was introduced.18 The authors concluded that if the U.S. was going to implement an anticipated rotavirus vaccine program, increased hospital surveillance would be called for because we didn’t have good data.19 Many pathogens are associated with the same symptoms as rotavirus (like norovirus or influenza), so it’s not possible to diagnose a child simply by looking at them. Lab testing is necessary to confirm the cause of the illness. This is significant because if a country is not lab testing for rotavirus, the burden of the illness and the potential usefulness of a vaccine are unknown. But the U.S. was ready to track the data because the World Health Organization had just added a new code for diagnosing rotavirus diarrhea in 1992, (If you don’t know, the WHO owns the codes used in health care around the globe). As fate would have it, a rotavirus vaccine just happened to be on the horizon.

And in 1998, the year the vaccine became licensed, many of those CDC authors published an updated assessment of rotavirus presence in diarrhea cases in the hospital, concluding that there were enough to “underscore the need for vaccinations.”20
But something else was brewing: a newly emerging global health philanthropic stew. The 1996 NEJM paper showing that natural immunity to rotavirus extinguishes reinfection came at a serendipitous time for science. A big player was about to move onto the field who would take up rotavirus vaccines and run. The new VIP was computer scientist and businessman Bill Gates, and he points to Bishop’s discovery of rotavirus – and the ensuing vaccine development – as “spur[ring] the way for GAVI,”21 the Global Alliance for Vaccines and Immunization.
Gates recalled that reading a 1997 New York Times (NYT) article about diarrhea deaths in third world countries changed his life. To paraphrase his words, he learned about a problem that seemed like it’d already been solved in rich countries. Kids were “being exposed to… dangerous pathogens that spread easily where toilets are scarce and water is contaminated.” So with his billions of dollars, he rushed past the logical steps of addressing sanitation and hydration, or supporting breastfeeding, and leaped right into funding vaccine research.

GAVI was created in 2000 amid the rise of public-private partnerships in the 1990s and 2000s. GAVI replaced a Children’s Vaccine Initiative (CVI) run by the UN, the WHO, the World Bank, and the Rockefeller Foundation meant to increase vaccines in developing countries.22 The powers involved with the CVI “recognized at the outset that no single agency or organization has the resources and capabilities to achieve the goals of the CVI.” A cynic might observe that a public-private partnership could provide more money with less red tape.
Gates reminisced on gatesnotes.com: “Melinda and I always had plans to do philanthropy, although much later in our lives. But after we learned about rotavirus, it seemed like there was no time to waste. We started making grants in global health, leading to the creation of our foundation in 2000. Some of our foundation’s first grants went to support the development of an oral rotavirus vaccine.”

RotaShield: the first rotavirus vaccine
“The decision to develop rotavirus vaccines was predicated on the extensive burden of rotavirus disease among children worldwide.”23
The fact that rotavirus has an RNA genome that is segmented is a biologic fact that can be exploited in a laboratory to “intentionally reassort rotavirus strains and prepare candidate vaccine strains that incorporate desirable phenotypic characteristics of different parental viruses.”24 The rotavirus vaccine is called a “reassorted” vaccine for this very reason. It includes mixed-up genetic information from both animals and humans.
Scientist Albert Kapakian at National Institutes of Health (NIH) created the human-rhesus (monkey) reassortant vaccine. This means the vaccine strain for RotaShield was created in a lab by putting human and monkey viruses together and letting them replicate, setting the stage for “reassortment,” meaning (in oversimplified terms) they got mixed up intentionally. The scientist can then screen for the desired shuffled virus strain they created, isolate it, and replicate it. Another routine vaccination is made this way: FluMist, which was approved for at-home administration in 2024.25
In short, RotaShield was a live, oral vaccine that uses pieces of monkey viral genetics and is unavoidably contaminated with monkey fetal cells, an antibiotic, and an antifungal.26 The parent rotavirus strain was from a monkey with diarrhea. That sample was “passed” through monkey kidney cells and then monkey fetal cells, then grown in the monkey fetal cells. The RNA in the vaccine-strain is “reassorted,” meaning pieced together from both monkey and human viral gene segments. The CDC states that “[t]race amounts of fetal bovine serum, neomycin sulfate [an antibiotic], and amphotericin B [an antifungal] are present in the vaccine (less than 1 μg per dose).”

As is standard practice, the NIH Office of Technology Transfer sold a license to the pharmaceutical company Wyeth to scale up for manufacture.27
Safety signals
After the licensure of the vaccine in the United States, approximately 600,000 infants were given RotaShield.28 In the initial recommendations, the CDC advised against vaccination after 7 months of age because of the risk of fever in older infants. “Vaccination should not be initiated for children aged greater than or equal to 7 months,” the CDC stated, “because these older infants might have an increased risk of fever occurring 3-5 days after receiving the first dose of vaccine”29
RotaShield was licensed on August 31, 1998, and recommended by the CDC on October 22, 1998. Immediately, there were reports of intussusception (when the intestine folds in on itself) after vaccination. One retrospective study published in 2001 using electronic health data from 10 managed care organizations found the risk to be 1 in 11,073 children vaccinated.30
The U.S. has more than one vaccine surveillance system: Vaccine Safety Datalink and VAERS. The first reports showed up in the Vaccine Safety Datalink (a collaborative database of electronic health records from specific insurance companies), followed by VAERS, the Vaccine Adverse Event Reporting System (a voluntary reporting system for anyone). The “Rotavirus Intussusception Investigation Team (RIIT)” was formed and quietly started their investigation on May 27, 1999, about seven months after the vaccine rollout, after nine cases were reported to VAERS. 31

In July 1999, the CDC published an official notice saying a safety signal had been found, and they would “pause” their recommendation, anticipating more cases.32
In reviewing the clinical trials, the investigators cited a publication from 1998 titled “Lack of an apparent association between intussusception and wild or vaccine rotavirus infection,” which reported that in pre-licensing vaccine trials, there were five cases of intussusception among 10,054 vaccinated infants, compared to one in 4,633 infants who got the placebo. The authors of that study, as you can tell from the title, did not find that difference to be significant and concluded there was no proof the vaccine caused the injury. Clearly, they were wrong.

Contrary to the 1998 publication, the RIIT found a “strong association between vaccination with [the vaccine] and intussusception among otherwise healthy infants [that] supports the existence of a causal relation.”33
The authors also found that “a higher proportion of the infants with intussusception were male and were Hispanic or black. The mother’s level of education was lower among these infants, they more often had Medicaid health coverage, and they less often had started consuming solid food before the reference date.”
The RIIT team also found their data confirmed previously understood protection of breast milk –it “modified the risk of intussusception” when babies got the vaccine. Interestingly, the investigators also reported, “Data from prelicensing trials with candidate rotavirus vaccines suggested that replication of [vaccine-strain rotavirus] is lower among breast-fed infants.” In other words, when the baby is breast feeding, the vaccine-strain pathogen can’t replicate as much, and in turn, the baby is less likely to be injured from the vaccine.
Other safety considerations: “In 2010, “porcine circovirus” – a pig virus that can disrupt growth and cause stillbirths – was detected in both rotavirus vaccines.” But we are assured, “There is no evidence that this virus is a safety risk or causes illness in humans.” 34
How was RotaShield licensed despite known safety risks?
In short, there were failures of judgment at both the FDA and the CDC that brought RotaShield to market.
Step 1: FDA licensure
The first hurdle a vaccine manufacturer has to jump in order to bring their liability-shielded product to market is the FDA. The department within the agency responsible for vetting and licensing vaccines is the Center for Biologics Evaluation and Research (CBER). When you hear a vaccine is “safe and effective,” it’s CBER’s stamp of approval Americans are relying on.

The FDA’s advisory committee tasked with reviewing vaccines is the Vaccines and Related Biological Products Advisory Committee (VRBPAC). VRBPAC’s December 1997 meeting was dedicated to the RotaShield licensure vote. One member raised concerns about intussusception that had been raised in other advisory committee meetings.35 Dr. Rennels, of the U.S. rotavirus efficacy group, presented the safety and efficacy results of a national multicenter and was rushed through her answer. She said that though there was intussusception seen in the trials, there was “no significant differences in the rates” between vaccines and placebos. She did admit she “was concerned that with larger numbers perhaps a causal relationship might emerge.” She wanted to talk about that for five minutes, but was limited to one or two. Rennels explained that she and FDA scientists investigated literature and concluded the intussusception in the trials was “chance temporal association” – in other words, a coincidence.

That was the only discussion of intussusception at VRBPAC before the unanimous yes vote to license RotaShield, despite the fact that the FDA is responsible for the safety and efficacy of licensed pharmaceuticals, and that supposedly well-researched opinion is relied upon by the CDC, doctors, policymakers, and parents.
The FDA is supposed to be the agency where vaccines are deemed “safe,” yet there was limited time for discussion, and no more questions about what turned out to be a serious and predictable problem. “Intussusception was not among the adverse reactions listed in an FDA press release issued upon the vaccine’s licensure, nor was it among the specific focus areas for postmarketing studies that Wyeth was instructed by the FDA to conduct.”36
Step 2: CDC recommendation
The next stop for vaccine makers is the CDC – more specifically, AICP, the Advisory Committee on Immunization Practices. Their recommendation will make or break a vaccine. Despite the fact that their recommendations do not have the force of law, they do end up being adopted in states that make them law, and federal and insurance payments are conditioned on following the schedule. As an investigation called by former Congressman Dan Burton put it: “The recommendation for routine use of a vaccine is tantamount to a federal mandate for vaccine use.”
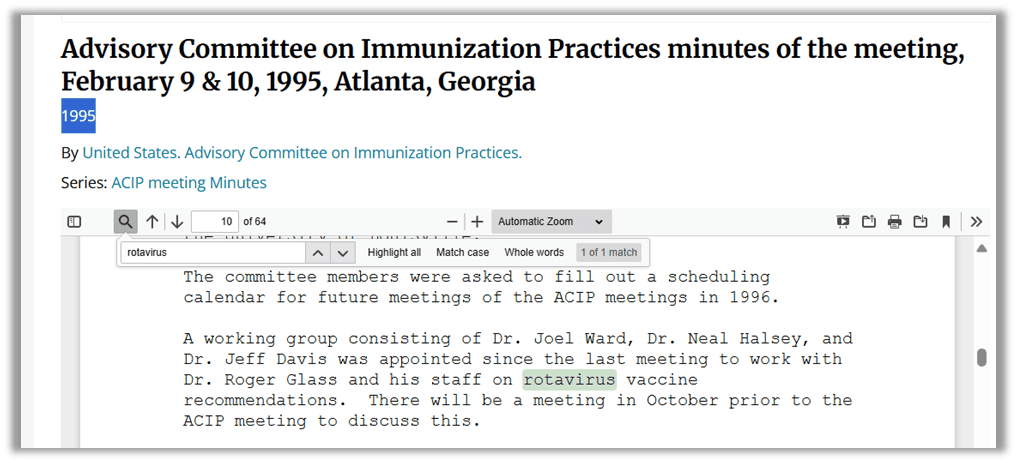
The ACIP started discussing the shape of recommendations for the rotavirus vaccine at least three years before it was licensed. A working group was created in 1995 to “work on rotavirus vaccine recommendations.”37
It is highly unusual that the ACIP would determine a recommendation for a vaccine before it was licensed by the FDA. Veteran vaccine expert, former member of and VRBPAC, consultant to Pfizer, vaccinologist, pediatrician, and federal government worker Kathryn Edwards wrote an article in 2025 bemoaning the changes to COVID shot policy recommendations made by Secretary Kennedy and asserted: “The usual sequence is that the vaccines are licensed by the FDA, and then the ACIP issues guidance based on a defined set of considerations.”38 Few people on the planet would know the system better than she does.
The ACIP determined before the vaccine was licensed that they would recommend it. This prelicensure reassurance was confirmed by one of the creators of the vaccine:39

The full ACIP panel had one of their first open meetings on the rotavirus vaccine in February 1997.40 The minutes from the meeting state, “After FDA approval, they recommend incorporating this vaccine into the vaccination schedule for infants” (emphasis added).
But how many doses? Even though evidence showed two doses were enough, the ACIP aligned with the WHO in saying there should be consistency with other vaccines, which is a strategy to get more vaccines in more kids. So rotavirus vaccines were recommended to be given at the same time as the other vaccines given at 2, 4, and 6 months of age.

Coincidentally, at the October 1997 meeting, the ACIP voted to change the minimum recommended dose schedule for polio vaccine’s third dose from 12 months to 6 months of age.41 It just so happened they wanted to “harmonize” the polio schedule with a newly approved polio vaccine, but that new recommendation also brought CDC recommendations in line with the WHO suggestion to bundle rotavirus right in there. Timing shots together is a common policy strategy to increase vaccine uptake; it isn’t a medical or health decision. This was highlighted in the HPV story where ACIP chose to recommend HPV for pre-teens getting other shots rather than 9 year olds the drug was approved for, and you can see it in the well-child check-ins with pediatricians.
“Simplifying” the CDC recommended schedule is a strategy the ACIP has been discussing since its beginning. In 2020 it became a tried and true strategy for increasing vaccine uptake: bundle the immunizations together, recommend the timing of the shots or drops be at the same time as “routine” pediatric care (like the well-visit schedule). This strategy of timing shots with regular visits is so well known it has been openly discussed by industry insider and former FDA Director Scott Gottleib on national news.42
The ACIP was fully aware of the intussusception injury and, in fact, discussed it far more in public record than VRBPAC or the FDA did. From an ACIP meeting June 1997, Discussion of intussusception in clinical trials:

Also note, from the same meeting, the intussusception happened up to 51 days after vaccinations.

This is highly relevant because any parent who would want to petition the government for compensation through the Vaccine Injury Compensation Program (VICP) will have a much harder time if the injury happens almost two months after the vaccination. Most acknowledged potential injuries are only considered compensable without a (big) fight if they happen within hours of the vaccine. Only a small handful of injuries are considered a result of a vaccine after a month and none are considered after 42 days. Rotavirus vaccine injuries are only considered if they happen within 21 days of the vaccine being given.
Despite all this information, the ACIP Rotavirus Working Group determined that the best recommendation the ACIP could make was to simply vaccinate all U.S. infants between 6 weeks and 6 months of age, in a one-size-fits-all dictate. The working group had the following options: recommend for all infants, allow for all infants, or select a high risk group of infants (or any combination of those options). It was determined that high-risk kids were too difficult to identify, and “permissive” recommendations (saying parents “can” rather than “should”) wouldn’t convince enough parents to take their kids in for shots, so the universal recommendation saying all kids should get the shot was the best option. So the policy became one-size-fits-all.
Here’s a list of specific considerations the Rotavirus Vaccine Working Group discussed and presented to the ACIP and, in each case, decided there was no safety concern, so they should recommend vaccination for all infants (with maybe a precautionary note in the small print of the vaccine info):
- Should we vaccinate preemies? Yes. There “was very little data on premature babies,” but a “theoretical concern” that it was “risky.” It was reiterated that “there’s still no good data on vaccine efficacy or safety in premature babies.” Still, there was a “general feeling that premature babies probably are at a higher risk for rotavirus hospitalization,” therefore, preemies would be grouped in with everyone else, but there would be a “word of warning.”
- Should we vaccinate around the immunocompromised despite shedding? Yes. It was determined that infants in households with immunocompromised people should get vaccinated because there was “nonexistant” data on whether the vaccine virus would cause illness, and there was risk of the immunocompromised person falling sick from a wild-type rotavirus.
- Should we vaccinate when babies are getting other blood products with antibodies? Yes. “There’s no data on the efficacy or the safety of rotavirus vaccine administered with or without… or the timing near antibody-containing blood products.” The working group opted to stay with the one-size-fits-all recommendation because otherwise, “you’re going to lose those kids to vaccination, and in which case, go ahead and do it.” The justification was, “again, lacking any data, but knowing” other vaccines are administered simultaneously with antibody-containing blood products, it was a go.
- Should we vaccinate despite knowing injuries happened in clinical trials? Yes. There was “a lot of discussion” about the potential for intussusception (the injury that ultimately got the vaccine booted from the schedule), and failure to thrive, but the concerns weren’t enough to stop a universal recommendation for all babies starting at 6 weeks old.
- Should we vaccinate kids with gastrointestinal disease? Yes. In initial drafts, pre-existing gastrointestinal disease in infants was a contraindication (meaning, don’t vaccinate these babies!). However, after “a long discussion…it was finally decided to allow vaccination of these children, but provide – again, not a precaution – but a special situations note at the end regarding the fact that there really is no safety and efficacy data in these children,” and so instead of employing the precautionary principle, erring on the side of caution, and limiting recommendations, it was determined to go ahead since there was no data to say they shouldn’t.
How many parents were informed about these discussions before giving the OK for their precious babies to be given the rotavirus vaccine? How many doctors? Was anyone told safety was determined by the lack of data, and an assumption that “no news is good news,” rather than collection of data affirming safety? Did they know there was “a lot of discussion” of known safety risks and concerns before the one-size-fits-all universal recommendation was made?

Ironically, one ACIP member piped up to mention a pattern he saw with their recommendations: He cautioned that each time they universally recommended a vaccine, “there is usually something that’s found out within a couple of years that we didn’t anticipate at the time.” He was talking about monetary costs, but that statement can clearly be applied to safety. He voted yes to the universal recommendation.

RotaShield became a scapegoat to stop vaccine hesitancy
RotaShield was licensed in the U.S. in 1998 and soon after recommended by the CDC. What else was happening in the world of vaccines during that time? Andrew Wakefield caused an earthquake in vaccine confidence by suggesting the MMR be split into 3 shots for safety precautions; the FDA and American Academy of Pediatrics jointly recommended thimerosal be removed from all vaccines for safety precautions;43 and Congressman Dan Burton was holding a public series of hearings on vaccine safety and policy to expose a system that he blamed for causing autism in his grandson.
As noted by medical ethics researcher Jason Schwartz, “The multifront attack on vaccine safety in 1999 led senior CDC officials to believe that without decisive action to preserve public support for vaccines and vaccine policymakers, the entire U.S. immunization program would be at risk.”44
“Jeffrey Koplan and Walter Orenstein knew that their critics were alleging that the CDC cared more about vaccine promotion than vaccine safety, and RotaShield was a vaccine with a known, serious risk.”45 It wasn’t hard to prove like autism and thimerosal claims were. Further, the manufacturer Wyeth was an easy target since they had just had their diet drug fen-phen taken off the market after people realized it caused heart problems.46
Wyeth saw the writing on the wall and did brand damage control. “Wyeth, recognizing from its communications with the CDC that the vaccine’s fate was sealed, preemptively withdrew RotaShield from the market on October 15, 1999, one week before the ACIP’s scheduled deliberations.”47
In response to the RotaShield scandal, the Committee on Government Reform, headed by Burton, did an investigation into conflicts of interest on the part of federal policymakers, specifically at ACIP and VRPBAC. The report was issued on June 15, 2000, and a hearing was held in Congress.48 (This is the report referenced by HHS Secretary Robert Kennedy when he fired all 17 members of the ACIP in 2025.49) The scathing report states: “Perhaps one of the major problems contributing to the overall influence of the pharmaceutical industry over the vaccine approval and recommendation process may be the loose standards that are used by the agency in determining whether a conflict actually exists… In many cases, significant conflicts of interest are not deemed to be conflicts at all.”
The actual conflicts of interest for both the ACIP and VRBPAC were detailed in the document and are too numerous to list here. The source document is highly recommended reading.

In his opening statement at the hearing, Burton pointed out, “Members who can’t vote in the full committee because of conflicts of interest are allowed to work on the same issues in working groups, and there is no public scrutiny.”50 The working groups “meet behind closed doors, out of the public eye.” Burton stated he was “appalled to learn that at least six of the 10 individuals who participated in the working group for the rotavirus vaccine had financial ties to pharmaceutical companies developing rotavirus vaccines.”
The investigation found that the CDC granted ACIP members “blanket waivers” for the entire year, which created “an environment where people do not take the conflict of interest issue as seriously as they should.” ACIP members were allowed to vote even when they had financial ties to competing companies making the same vaccine. This is a problem because recommendation of one brand opens the door securely for any competitor. This was the case for Paul Offit, who was working with Merck to develop RotaTeq when he voted yes on Wyeth’s RotaShield as a new member of ACIP. He was off the ACIP by the time of the RotaTeq vote, but that initial “yes” paved the way for his vaccine.
To think the “experts” didn’t know what they were doing would be naive. In the June 1998 meeting, the ACIP chose to have an attorney present for questions about ethics and conflicts, again before RotaShield was licensed by the FDA. The chair of the committee asked if members who had stock in Wyeth, the maker of the vaccine, could vote. How could a person esteemed to be a world expert on vaccines, chairing a panel of experts, have such a confusion about whether there is a conflict about voting on a product when you can benefit from its success? The ACIP attorney came from the same school of thought apparently, calling it a “gray” area that requires discussion because the “FDA has an incredibly complicated process.” To his credit, the attorney claimed he would “personally recuse [him]self from voting,” if they felt there was a potential conflict.

In that response, the attorney pointed out something important: ACIP is not the same as the FDA’s VRBPAC. The ACIP mainly exists to handle votes about the Vaccines for Children (VFC). That matters because VFC controls government vaccine purchasing agreements and also when the ACIP votes to add a new childhood vaccine (or a similar product) recommendation to the VFC list, that vaccine becomes shielded from lawsuits.
ACIP exists to make kids get more vaccines
Anyone who has endured even a handful of ACIP meetings knows that cost is always a discussion, typically painted in the “benefit” column of a risk benefit analysis.


Why would the ACIP consider the cost of a medical product in deciding whether all children in the U.S. should get it? Isn’t this a public health committee? Should the people considering whether a vaccine is safe and effective enough to recommend for all of our babies be concerned about the cost?
The lawyer let the cat out of the bag – cost is a decision factor because ACIP is “an operational group for the purposes of VFC votes.” They’re a VFC machine, not a safety check. Despite any safety concerns or discussion that arises for public record, members typically vote yes regardless of their reservations and questions. We can give them the benefit of the doubt and say, in theory, the FDA has checked safety and efficacy, and the ACIP is going to lean on that. Assuming the FDA did its job right, the ACIP’s role is reading the room about how American parents are going to react to another addition to the schedule and determine the best way to convince parents their kid needs more doses. Its focus is messaging and justifying costs to the American people. If policy discussion is obscured by conversation about made-up cost savings, one more pharmaceutical on the schedule is an easier pill to swallow.

ACIP Secretary Dr. Dixie Snider stated: “The overall goal of the ACIP is to provide advice that assists CDC, HHS, and indeed the whole nation, in reducing the incidence of vaccine preventable diseases and increasing the safe usage of vaccines and related biological products.” ACIP is there to maximize vaccine usage. Full stop.
It’s right there in the name. The Advisory Committee on Immunization Practices rests on an underlying assumption that vaccines are the way to stop infectious disease… or at least that’s what the point used to be, but in our post-COVID world, vaccines are about taming and lessening sickness, not prevention. The ACIP is not a place where vaccines as a public health policy are debated or questioned. The only question they are concerned with is ‘how do we increase vaccine usage.’ From the outside looking in, we may think that it’s reasonable for the ACIP to wonder if a vaccine is needed at all. Wouldn’t the necessity of a vaccine be a part of a risk-benefit analysis? Not at ACIP. The ACIP has marching orders to advise on distributing vaccines, not deciding if they’re needed. The science and statistics that are paraded out before a vote may look like a safety check, but in practice, they aren’t. It’s assumed and agreed upon that risk is acceptable for “the greater good.” Their discussions of safety are limited in sample size and what they screen for and are more appropriate for assessing what the public knows about risk and what will be tolerated.
The ACIP’s role is to anticipate public perception and stop vaccine hesitancy.
The ACIP knew before recommending the rotavirus vaccine that American parents wouldn’t see a need for it. At an ACIP meeting in June 1997, one of the members remarked: “[T]here is a potential problem of the perceived need for this vaccine, which I think needs to be addressed. If we do have a vaccine, if we do go with universal immunization, we will have, we really do need to embark on a large-scale education program for practitioners and the general public” (emphasis added).
This is why, even before the vaccine was licensed, the ACIP started talking about it and had zeroed in on the messaging and “target” for the recommendation in the U.S.51 The publicly stated goal wasn’t to reduce deaths, but to reduce hospitalizations by 3-4% and doctor visits by 20-30%. That’s it.

In the same statement, the speaker noted they knew about the chance of intussusception and fevers strong enough to be reported in vaccine clinical trials.

To sum up: Before the vaccine was licensed, our best and trusted experts knew about intussusception and high fever risks and failure to thrive injuries. They weighed that against reducing hospitalizations by only 3-4%, or normal sick visits by up to 30%… and ultimately said, yes, that’s worth a recommendation for all American children. They didn’t stop and say, hey, maybe American parents won’t want this. Maybe we should be extra clear that there are risks, and we should recommend “shared decision-making” so parents and doctors can assess together whether this is right for a child. No. Instead they buried the information and had discussions about how to “educate” and use the right messages, including how to combat resistance when parents learned about the intussusception risk.
Is that a decision you would make for your baby? Especially knowing that hospitalization for rotavirus is more likely in certain areas of the country, most likely in another country, and can be prevented by proper nourishment and hydration?
The CDC and the FDA were wrong about intussusception risk with RotaShield.
And it turns out they were wrong again when the second generation of rotavirus vaccines were approved and put back on the schedule.
Rotavirus vax is back
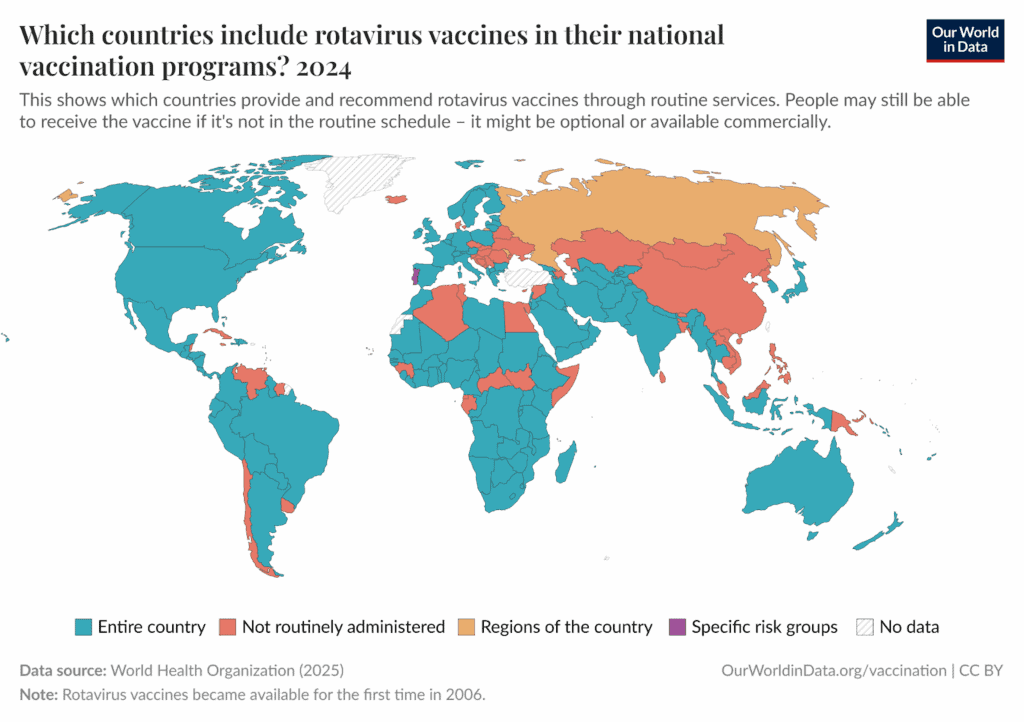
The removal of the rotavirus vaccine from the childhood vaccine schedule was not intended to be permanent. There were other manufacturers with rotavirus vaccines in the pipeline, waiting to move in. The World Health Organization and GAVI were seeking a new candidate. Eventually and inevitably, companies filled the void with new vaccines.52 Regardless of the well-known deficiency in developing countries, the WHO greenlighted them worldwide, and the ACIP put it right back on the schedule for American infants too.
In 1999, the Wyeth RotaShield’s fate was sealed beyond U.S. development. Countries around the globe look to the U.S. CDC as a gold standard for public health. After the ACIP deemed the vaccine unsafe for American children, it couldn’t be sold in other countries because it was publicly rejected. The ACIP knew the power and politics of their decision to remove it and even had discussions about how they would frame their announcement and withdraw so as to not affect the global market! Yet again, their focus was on protecting the vaccine program rather than people, by marketing and controlling the narrative. But global leaders in developing countries were not having it. At a WHO-hosted meeting on the future of the vaccine in February 2000, public health ministers from other countries told Americans “they could not ask their citizens to accept a vaccine deemed too dangerous for American children, even in countries where as many as 1 in 250 children died from rotavirus-related illness.”53
Stanley Plotkin, lead author of “Plotkin’s Vaccines,” which is akin to the Bible for vaccine religion, reflected on this refusal to push a dangerous drug on babies with a comment revealing his priorities: “No country was willing to place public health above possible criticism for using a vaccine rejected by the United States. This was not exactly a profile in courage.”54 He reduced the documented risk from the dangerous vaccine to “possible criticism” from citizens if that risk was accepted and promoted. Plotkin literally wrote the book on vaccines and is known as the “Godfather of Vaccines.” He is revered as a leading expert, and the ACIP has started every meeting since 2018 with a gavel engraved with his name. This shows us clearly that safety is something each parent needs to be aware of and assess; it is not to be left to vaccine “experts.”
It also happened that Plotkin was one of the co-inventors of the competitor rotavirus vaccine made by Merck, alongside Paul Offit and Fred Clark. It is one of two rotavirus vaccines licensed in the U.S. The other is Rotarix.
Paul Offit, Stanley Plotkin, and Fred Clark were given a grant by Merck to develop a rotavirus vaccine. Offit was on a scientific advisory board for Merck while serving on the ACIP and when he voted yes for RotaShield. In 2005, Merck funded a “Maurice R. Hilleman Chair of Vaccinology,” which was awarded to Paul Offit. That is 20 years of benefitting from Merck’s funding. In 2008, CBS News reported that Chair was worth $1.5 million55. Offit admits to profits of $6 million from the sale of the patent.56 (Some critics assert this is not the whole picture and his profit was much more.)57

Rotavirus vaccines still cause intussusception
Shockingly, the reference book for infectious disease, the AAP Red Book, states, “Postmarketing surveillance data from the United States, Australia, Mexico, and Brazil indicate that there is a small risk of intussusception from the currently licensed rotavirus vaccines. In the United States, the data currently available suggest the attributable risk is between approximately one excess intussusception case per 20,000 to 100,000 vaccinated infants. …In the United States as well as other parts of the world, the benefits of rotavirus vaccination in preventing severe rotavirus disease outweigh the risk of intussusception. Parents should be informed of the risk…” (emphasis added).
The AAP may call it a “small” risk, but the World Health Organization notes that this vaccine has a “good safety profile, but that it may be associated with an increased (up to six-fold) risk of intussusception after the first dose of vaccine in some populations.”58
In fact, intussusception is still recognized as a vaccine injury by the VICP in 2025.59 This is notable because despite the wide range of potential injury from vaccines, very few are so well known and irrefutable that they make it to the “Table,” which is a very short list of injuries from vaccines acknowledged by our government. If a vaccine injury is on the Table, a parent shouldn’t have to fight the government on what caused a child’s health problems, disability, or death.


The package insert for Rotarix summarizes the warning: The risk of intussusception from this vaccine still exists even though it was put back on the schedule. It’s just a different risk at a different timeframe: “Among vaccine recipients, there were no confirmed cases of intussusception within the 0- to 14-day period after the first dose… which was the period of highest risk for the previously licensed oral live rhesus rotavirus-based vaccine.”60 But that was just one of their studies in Latin America and Finland. In other studies, in Mexico and Brazil and Australia, there was a “temporal association between vaccination with Rotarix and intussusception” within 31 days, with a cluster in the first week, directly contradicting the info from the Latin American study and putting Rotarix squarely on par with RotaShield.

A 2016 study mentioned a “slightly higher risk,” but didn’t explain how quickly health officials detected it, even though they were supposed to be watching closely.61 In large clinical trials with over 60,000–70,000 babies, no link to intussusception showed up. But once the vaccines were released, follow-up data revealed a small increase in cases. This happened even though the FDA and the WHO told researchers to look out for that specific problem. In the end, they found about one to six extra cases of intussusception for every 100,000 vaccinated infants.
Another study claimed about one to two extra cases of intussusception per 100,000 vaccinations, about one tenth the risk seen with the older Rotashield vaccine.62 But the researchers admitted the real risk might be higher, possibly as much as one case for every 31,000 shots. All of these figures are based on statistical estimates rather than exact counts.
No matter how you look at it, the risk of intussusception from a rotavirus vaccine seems higher than the often-repeated claim that vaccine injury risks are only about one in a million.
So are the current vaccines safer than the last? And by how much? What level of risk is enough to say a vaccine is not safe? In 1999 it was 1 in 10,000 babies…in 2025 it appears 1 in 20,000 is deemed acceptable risk.
Adding insult to injury, the vaccine is less effective in poorer countries
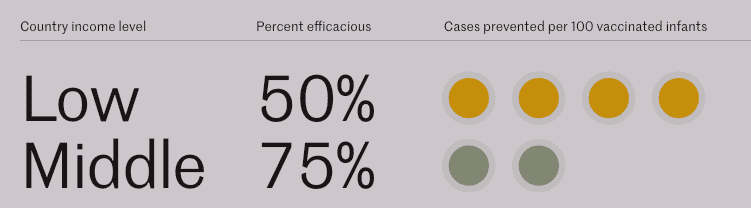
The RotaShield vaccine was tested in the United States and Finland, despite the impetus for the vaccine being for developing countries. Based on the data, the ACIP reported that the vaccine prevented 49% to 68% of all rotavirus diarrhea cases, 69% to 91% of severe cases, and 50% to 100% of doctor visits for rotavirus treatment.63 And remember, as we pointed out in Shining a light on HPV, the efficacy in clinical trials is always higher than what happens in the real world, so those numbers would only go down.
Unfortunately, the rotavirus vaccine worked even less effectively in the very countries it was designed to help. “Rotavirus vaccines have also been shown to be 23% to 47% less effective in high child mortality regions compared to low child mortality regions.”64 And this was expected because experts have long believed that vaccines usually produce stronger immune responses in children from developed countries compared to those in developing countries, presumably because of nutrition differences.65
Some scientists suggest that giving the vaccine earlier might make it work better because it could change how the baby’s gut develops. The gut clearly matters, since breastfeeding helps protect against both infection and vaccine injury. The vaccine is also least effective in developing countries, where malnutrition is widespread.66
Supporters of the vaccine argue that, despite being less effective in poor countries, rotavirus vaccines actually make a bigger difference there than in wealthier nations.67

They argue that because poor countries have far more rotavirus cases, even a less effective vaccine prevents more total cases than in wealthy countries. This is used to counter the fact that the vaccines barely work. But there’s no evidence that lower effectiveness also means lower risk.
Remember, we were sold on the rotavirus vaccines because so many children around the globe are dying. And since the vaccine is needed in developing countries, we may as well use it here, right? In discussions before the vaccine was licensed, the ACIP noted the vaccine would have the “greatest impact” in developing countries.

But, remember, which country was the first to license and roll out RotaShield?
It was the United States.
It was introduced in the U.S. first because we would pay for it, and the cost would be lower in other countries.68 That’s by design. It’s “classical vaccine development,” where companies introduce a new vaccine in developed countries at high prices, then sell the vaccine to underdeveloped countries at a reduced cost.69
The experiment on American children, meaning the widespread understanding of a problem only after we started giving it to our children en masse, stopped the rollout in any other country until the new vaccines were introduced in 2006.70
The doublespeak about how the vaccine works continues in an article cited by Paul Offit and others in their chapter on rotavirus for “Plotkin’s Vaccines.” The vaccine won’t be cost-saving in the United States, meaning we’ll pay for it and it won’t save us money down the line in overall health care costs in the country. It’s just a straight-up expense. Most vaccines are justified by saying that we’ll pay now, but we’re going to save so much money in the future by avoiding expensive hospitalizations and lost wages from parents having to take time off work. Not for RotaShield. But Offit assures us it will be cost-effective anyway, which is health-speak lingo that means they’ve decided the benefits outweigh the cost.71

The ACIP had estimated the vaccine would only be cost saving in the health system if it were $8 or less per dose, and would yield savings in “societal costs” if it was under $41. Rotashield was listed for $38 per dose, and ACIP hoped they would sell it to the federal government for less.72
Adding even more insult to injury here is the fact that the vaccine made the virus worse in the wild. As we have seen with many of the vaccines, rotavirus is a moving target. Introduction of a vaccine into a field where the target can mutate creates pressure that drives strain replacement. In other words, the vaccine changes the illness which, in turn, changes whether and how the vaccine will work.

Future of vaccine production
Recent data estimates about 75% of U.S. children complete the rotavirus vaccine series. Around 3.6 million children are born yearly in this country, so around 2.7 million U.S. children are given rotavirus vaccines to try to avoid an illness that can be asymptomatic, only rarely requires hospitalization, and is easily treated. For that lottery, they risk an injury that would require hospitalization with surgery instead of just fluids.
Still, science celebrates this vaccine, despite its failures. Think about this statement from a 2008 publication titled, “Why does the world need another rotavirus vaccine?” The article is an homage to the 60-year process, the tens of thousands of babies, and the billions of dollars spent on the “successful” vaccines… and a look toward alternative vaccines and why they’re needed.

The work started by Bishop when she discovered rotavirus continues today. The University of Melbourne has been working on developing a rotavirus vaccine to be administered at birth before 4 weeks old (referred to as “neonatal”), with the hopes that immediate dose would “overcome challenges to vaccine uptake posed by a complex gut microbiome.”73 Indeed, research published in 2025 showed that dosing infants would change the development of their gut flora at a critical time in their early development. Scientists hope by altering this critical development, babies will have a better response to the vaccine.
At birth, when children are breastfed, the protection from the milk stops a vaccine from working well in the baby. Breast milk makes vaccines less effective in infants. But this vaccine strain is unique because it is derived from an asymptomatic infant, so it can “replicate well in the presence of maternal and breast milk antibodies.”
Do you think scientists should try to find a work around for breast milk so we can alter the forming gut of an infant just so a pharmaceutical product can have a stronger effect on that baby?
Conclusion: VAERS works
So does VAERS work? It sure does – even with very well-known flaws that lead to underreporting by magnitudes of 10s. What’s broken is the response to the safety signals.
Compare intussusception numbers. They started investigating once nine reports were made, but look how many reports are in VAERS now! Why hasn’t the vaccine been pulled again? What other vaccines carry reports that reflect a 1/10,000 risk of injury, or even 1/100,000?
Neither the CDC nor the FDA have a testable, scientific or legal definition of safety. The FDA has stated safety is a “relative freedom from harmful effects.”74 It changes with each situation. It’s subjective. What this means is, the safety threshold is public perception. Will the public accept this vaccine or not? Are we going to ask questions? Will we stand up when we have doubts?
Rotashield was a scapegoat to take eyes off of the MMR and vaccine questions in general. Because of Wakefield and the Institute of Medicine questioning thimerosal, and the bravery of a sitting congressman to hold investigations on vaccines, in the late 1990s there was a “terrible storm where a growing number of doctors and devastated families were finding their own links between autism and vaccines, and governments wanted to destroy that mounting association with no questions asked.”
Orenstein stated in an interview in 2009, “This was not viewed as rotavirus simply for rotavirus’s sake but also the potential for maintaining public faith and credence in the overall immunization schedule.”75
It’s interesting to ponder, knowing VAERS is an incomplete picture, that there were 112 cases ultimately reported, 95 of which were confirmed by medical review by 2004. For a passive surveillance system, where we have continual reminders that the reports are “unconfirmed,” that 95/112 equals 84.8% confirmation rate, which is a pretty strong signal.
Regardless of their conclusion that the vaccine causes injury (which they knew from the clinical trials) the investigators on the CDC’s Rotavirus Intussusception Investigation Team parroted the talking point that the risk is worth the benefit of claimed protection against hospitalization from rotavirus.

Is that a position Americans should accept as public policy? When the CDC recommends a vaccine, even if there is an injury warning on the package insert, it reduces informed consent. Who should decide if the risk of an injury that requires surgical intervention is worth it compared to the risk of needing hospitalization to prevent severe dehydration?76 The ACIP knows that “in the United States most complications can be prevented by oral rehydration.” Which medical situation would you rather navigate with your child? What are the chances of either for your child? It’s surely not the same for every child, but the CDC seems to want our heads in the sand.
We are expected not to question whether the sacred cow called “vaccine” is safe. We are told over and over they are “safe and effective.” The government is vetting them! The government is watching them! Our surveillance systems work!
“[T]he RotaShield story is regularly invoked as evidence of the effectiveness of vaccine safety activities in the United States.”77 It was pointed to in the government’s support of stopping state actions against vaccine manufacturers for design defects in the notorious Brunsewitz v Wyeth, 562 U.S. 223 (2011), which severely narrowed the ability of Americans to pursue justice when they or their children are injured by these products.78 The sad story of RotaShield “can be found throughout the medical literature, in congressional testimony, and in public health planning documents.”
Medical ethics researcher Jason Schwartz put it succinctly: The legacy of RotaShield was reduced to “little more than ‘the system worked so the system works.’” Parents are expected to believe that since one vaccine was publicly raked over the coals and temporarily removed from the constantly growing schedule, that they can sleep soundly, believing the government wouldn’t let a dangerous product be given to our kids.
How many times have we heard, “We know VAERS works because of rotavirus”? This isn’t a talking point proponents of vaccines should be waving around. The story of Rotashield is the smoking gun used to kill informed consent for American parents.
And this is why Americans must never stop asking questions. As of July 2025, there are more than 2.6 million reports of adverse events in VAERS for all vaccines.
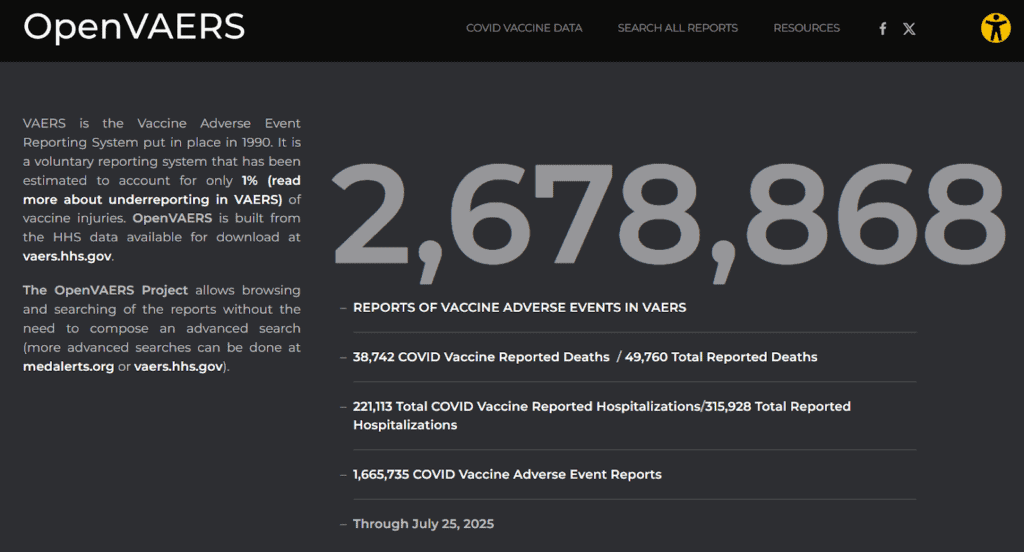
There are over 2.6 million injury and death reports, and only one small cluster of nine cases in seven months in the late 1990s prompted the CDC to (temporarily) reverse course and reassess the risks and benefits of a vaccine they recommended. There have been no others.
Why is that?
Here’s a statement to ponder, from Dan Burton in the June 15, 2000, hearing that focused on rotavirus failure: “If the panels that have made the decisions on all vaccines on the childhood immunization schedule had as many conflicts as we have found with rotavirus, then the entire process has been polluted and the public trust has been violated.”79
References
- Orenstein, Walter A., et al. 2023. Plotkin’s Vaccines. 7th ed. Elsevier. https://shop.elsevier.com/books/plotkins-vaccines/orenstein/978-0-323-79058-1, page 1008. ↩︎
- AAP Committee on Infectious Diseases. 2018. Red Book: Report of the Committee on Infectious Diseases. 31st ed. American Academy of Pediatrics. ↩︎
- Orenstein, Walter A., et al. 2023. Plotkin’s Vaccines. 7th ed. Elsevier. https://shop.elsevier.com/books/plotkins-vaccines/orenstein/978-0-323-79058-1, page 1005. https://publications.aap.org/aapbooks/book/546/Red-Book-2018-Report-of-the-Committee-on, page 700. ↩︎
- Orenstein, Walter A., et al. 2023. Plotkin’s Vaccines. 7th ed. Elsevier. https://shop.elsevier.com/books/plotkins-vaccines/orenstein/978-0-323-79058-1, page 1009.
↩︎ - Allen, Stephen J., et al. “Probiotics for Treating Infectious Diarrhoea.” Cochrane Database of Systematic Reviews, (2003). https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD003048.pub2/full.
↩︎ - AAP Committee on Infectious Diseases. 2018. Red Book: Report of the Committee on Infectious Diseases. 31st ed. American Academy of Pediatrics. ↩︎
- “Implications of Worldwide Population Growth For U.S. Security and Overseas Interests (THE KISSINGER REPORT).” National Security Study Memorandum, (1974). ↩︎
- Glass, Roger I., et al. “The Rotavirus Vaccine Story: From Discovery to the Eventual Control of Rotavirus Disease.” The Journal of Infectious Diseases, (2021). https://www.researchgate.net/publication/354997767_The_Rotavirus_Vaccine_Story_From_Discovery_to_the_Eventual_Control_of_Rotavirus_Disease. ↩︎
- Glass, Roger I., et al. “The Rotavirus Vaccine Story: From Discovery to the Eventual Control of Rotavirus Disease.” The Journal of Infectious Diseases, (2021). https://www.researchgate.net/publication/354997767_The_Rotavirus_Vaccine_Story_From_Discovery_to_the_Eventual_Control_of_Rotavirus_Disease. ↩︎
- “Ruth Bishop – The Rotavirus Story.” The Royal Children’s Hospital Melbourne. Video, https://www.youtube.com/watch?v=A9qG045viHE. ↩︎
- McDonald, Sarah M., and John T. Patton. “Assortment and Packaging of the Segmented Rotavirus Genome.” Trends in Microbiology 19, no. 3 (2011): 136-44. https://pubmed.ncbi.nlm.nih.gov/21195621/. ↩︎
- Orenstein, Walter A., et al. 2023. Plotkin’s Vaccines. 7th ed. Elsevier. https://shop.elsevier.com/books/plotkins-vaccines/orenstein/978-0-323-79058-1, page 1005. ↩︎
- Matthijnssens, Jelle, et al. . “Uniformity of Rotavirus Strain Nomenclature Proposed by the Rotavirus Classification Working Group (RCWG).” Archives of Virology 156, no. 8 (2011): 1397–1413. https://pmc.ncbi.nlm.nih.gov/articles/PMC3398998/#S2. ↩︎
- “The Impact of Rotavirus Vaccination.” Johns Hopkins University Bloomberg School of Public Health,. https://publichealth.jhu.edu/sites/default/files/2024-02/rota-brief7-vaccineimpact2022ax.pdf. ↩︎
- Cortese, Margaret M. MD, and Penina Haber MPH. “Chapter 19: Rotavirus.” CDC Pink Book, (2024). https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-19-rotavirus.html?utm_source=chatgpt.com. ↩︎
- Velázquez, F R., et al. “Rotavirus Infection in Infants as Protection against Subsequent Infections.” New England Journal of Medicine 335, no. 14 (1996): 1022-8. https://pubmed.ncbi.nlm.nih.gov/8793926/. ↩︎
- Velázquez, F R. “Rotavirus Infection in Infants as Protection against Subsequent Infections.” New England Journal of Medicine 335, no. 14 (1996): 1022-8. https://pubmed.ncbi.nlm.nih.gov/8793926/. ↩︎
- Glass, R I., et al. “The Epidemiology of Rotavirus Diarrhea in the United States: Surveillance and Estimates of Disease Burden.” The Journal of Infectious Diseases 174, Suppl 1 (1996). https://pubmed.ncbi.nlm.nih.gov/8752284/. ↩︎
- Glass, R I., et al. “The Epidemiology of Rotavirus Diarrhea in the United States: Surveillance and Estimates of Disease Burden.” The Journal of Infectious Diseases 174, Suppl 1 (1996). https://pubmed.ncbi.nlm.nih.gov/8752284/. ↩︎
- Parashar, U D., et al. “Hospitalizations Associated with Rotavirus Diarrhea in the United States, 1993 through 1995: Surveillance Based on the New ICD-9-CM Rotavirus-specific Diagnostic Code.” The Journal of Infectious Diseases 177, no. 1 (1998): 13-7. https://pubmed.ncbi.nlm.nih.gov/9419164/. ↩︎
- Gates, Bill. “This Virologist’s Discovery Saved Millions of Lives and Helped Launch Our Foundation.” Gates Notes, (2021). https://web.archive.org/web/20210318113307/https://www.gatesnotes.com/Health/Heroes-in-the-field-Dr-Ruth-Bishop. ↩︎
- Mitchell, V S., et al. “Why a Children’s Vaccine Initiative?” The Children’s Vaccine Initiative: Achieving the Vision, (1993). https://www.ncbi.nlm.nih.gov/books/NBK236423/. ↩︎
- Glass, R I., et al. “The Epidemiology of Rotavirus Diarrhea in the United States: Surveillance and Estimates of Disease Burden.” The Journal of Infectious Diseases 174, Suppl 1 (1996). https://pubmed.ncbi.nlm.nih.gov/8752284/. ↩︎
- Orenstein, Walter A., et al. 2023. Plotkin’s Vaccines. 7th ed. Elsevier. https://shop.elsevier.com/books/plotkins-vaccines/orenstein/978-0-323-79058-1, page 1005. ↩︎
- “FDA Approves Nasal Spray Influenza Vaccine for Self- or Caregiver-Administration.” U.S. Food & Drug Administration, (2024). https://www.fda.gov/news-events/press-announcements/fda-approves-nasal-spray-influenza-vaccine-self-or-caregiver-administration. ↩︎
- “Rotavirus Vaccine for the Prevention of Rotavirus Gastroenteritis Among Children Recommendations of the Advisory Committee on Immunization Practices (ACIP).” Morbidity and Mortality Weekly Report 48, ↩︎
- Rotavirus Vaccine for the Prevention of Rotavirus Gastroenteritis Among Children Recommendations of the Advisory Committee on Immunization Practices (ACIP).” Morbidity and Mortality Weekly Report 48, no. RR-2 (2000): 1-23. https://www.cdc.gov/mmwr/preview/mmwrhtml/00056669.htm ↩︎
- Khare, Meena, et al. . “Dynamics of RotaShield Uptake in the US Infant Population, a Vaccine that Was Withdrawn After 9 Months of Use.” CDC National Immunization Conference, (2002). https://www.cdc.gov/nchs/data/nis/miscellaneous/khare2003c.pdf. ↩︎
- “Rotavirus Vaccine for the Prevention of Rotavirus Gastroenteritis Among Children Recommendations of the Advisory Committee on Immunization Practices (ACIP).” Morbidity and Mortality Weekly Report 48, no. RR-2 (2000): 1-23. https://www.cdc.gov/mmwr/preview/mmwrhtml/00056669.htm. ↩︎
- Kramarz, Piotr, et al. . “Population-based Study of Rotavirus Vaccination and Intussusception.” The Pediatric Infectious Disease Journal 20, no. 4 (2000): 410-6. https://www.researchgate.net/publication/12003125_Population-based_study_of_rotavirus_vaccination_and_intussusception. ↩︎
- Murphy, Trudy V. MD., et al. “Intussusception Among Infants Given an Oral Rotavirus Vaccine.” New England Journal of Medicine 344, (2001): 564-572. https://www.nejm.org/doi/10.1056/NEJM200102223440804?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200www.ncbi.nlm.nih.gov. ↩︎
- “Intussusception Among Recipients of Rotavirus Vaccine — United States, 1998-1999.” Morbidity and Mortality Weekly Report 48, no. 27 (1999): 577-581. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4827a1.htm. ↩︎
- Murphy, Trudy V. MD., et al. “Intussusception Among Infants Given an Oral Rotavirus Vaccine.” New England Journal of Medicine 344, (2001): 564-572. https://www.nejm.org/doi/10.1056/NEJM200102223440804?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200www.ncbi.nlm.nih.gov. ↩︎
- AAP Committee on Infectious Diseases. 2018. Red Book: Report of the Committee on Infectious Diseases. 31st ed. American Academy of Pediatrics. https://publications.aap.org/aapbooks/book/546/Red-Book-2018-Report-of-the-Committee-on, page 702. ↩︎
- “RotaShield Rotavirus Vaccine Sponsor Presentation.” Vaccines and Related Biological Products Advisory Committee, (1997). https://wayback.archive-it.org/7993/20170404132536/https://www.fda.gov/ohrms/dockets/ac/97/transcpt/3360t2.pdf. ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- “Advisory Committee on Immunization Practices Minutes of the Meeting, February 9 & 10, 1995, Atlanta, Georgia.” CDC Stacks, (1995). https://stacks.cdc.gov/view/cdc/76596. ↩︎
- Edwards, Kathryn M. “The FDA Is Circumventing Key Roles and Procedures for Licensure and Use of COVID Vaccines.” STAT, (2025). https://www.statnews.com/2025/05/22/fda-covid-19-vaccines-novavax-acip-prasad-makary/. ↩︎
- Grantsome. (n.d.). Development and evaluation of live, attenuated rotavirus (Grant Z01-AI000341-20). Retrieved 10/1/2025, from https://grantome.com/index.php/grant/NIH/Z01-AI000341-20 ↩︎
- “Advisory Committee on Immunization Practices Minutes of the Meeting, February 12-13, 1997.” CDC Stacks, (1997). https://stacks.cdc.gov/view/cdc/76567. ↩︎
- “Advisory Committee on Immunization Practices October 22-23,1997 Meeting Minutes.” CDC Stacks, (1997). https://stacks.cdc.gov/view/cdc/76568. ↩︎
- “Dr. Scott Gottlieb on Why the Childhood Vaccine Schedule Isn’T Spaced Out More.” Chief Nerd. September 8, 2005. Video, https://x.com/thechiefnerd/status/1965148517107990665?s=46&t=I2wQOoBVjmDQKEo882eV7g. ↩︎
- Centers for Disease Control and Prevention. (1999, July 9). Notice to readers: Thimerosal in vaccines: A joint statement of the American Academy of Pediatrics and the Public Health Service. Morbidity and Mortality Weekly Report. Retrieved [date you accessed it], from https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4826a3.htm ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- Fauber, John , and Kristina Fiore. “Risk/Reward: Once Hailed as a Miracle Treatment, Fen-phen Became a Diet Drug Debacle.” Milwaukee Journal Sentinal, August 22, 2021. https://www.jsonline.com/story/archives/2021/08/22/once-hailed-miracle-treatment-fen-phen-became-diet-drug-debacle/5294734001/.
Cohen, Kate. “Fen Phen Nation.” Frontline, November 13, 2003. https://www.pbs.org/wgbh/pages/frontline/shows/prescription/hazard/fenphen.html. ↩︎ - Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012) https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- “Conflicts of Interest in Vaccine Policy Making.” Committee on Government Reform, U.S. House of Representatives, (2000). https://icandecide.org/wp-content/uploads/2023/01/OGR-Majority-Report-1.pdf. ↩︎
- Lyons-Weiler , James . “Former Vaccine Committee Did Not Follow the Rules.” Brownstone Journal, (2025). https://brownstone.org/articles/former-vaccine-committee-did-not-follow-the-rules/. ↩︎
- “FACA: Conflicts of Interest and Vaccine Development — Preserving the Integrity of the Process.” Committee on Government Reform, U.S. House of Representatives, (2000). https://www.govinfo.gov/content/pkg/CHRG-106hhrg73042/html/CHRG-106hhrg73042.htm. ↩︎
- “Advisory Committee on Immunization Practices February 11-12, 1998 Meeting Verbatim Transcript.” CDC Stacks, (1998). https://stacks.cdc.gov/view/cdc/76564. ↩︎
- Glass, Roger I., et al. “The Rotavirus Vaccine Story: From Discovery to the Eventual Control of Rotavirus Disease.” The Journal of Infectious Diseases, (2021). ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- Attkisson, Sharyl . “How Independent Are Vaccine Defenders?” CBS News, (2008). https://www.cbsnews.com/news/how-independent-are-vaccine-defenders/. ↩︎
- “Paul Offit Explains the Money Side of the Rotavirus Vaccine He Worked On.” Left Brain/Right Brain, (2009). https://leftbrainrightbrain.co.uk/2009/09/14/paul-offit-explains-the-money-side-of-the-rotavirus-vaccine-he-worked-on/. ↩︎
- Olmsted, Dan , and Mark Blaxill. “Counting Offit’S Millions: More on How Merck’s Rotateq Vaccine Made Paul Offit Wealthy.” Age of Autism, (2009). https://www.ageofautism.com/2009/12/counting-offits-millions-more-on-how-mercks-rotateq-vaccine-made-paul-offit-wealthy.html. ↩︎
- “Rotavirus Vaccines and Intussusception.” WHO Weekly Epidemiological Record, (2014). https://www.who.int/groups/global-advisory-committee-on-vaccine-safety/topics/rotavirus-vaccines/intussusception. ↩︎
- “Vaccine Injury Table Effective for Claims Filed on or After January 3, 2022.” Health Resources and Services Administration, (2017). https://www.hrsa.gov/sites/default/files/hrsa/vicp/vaccine-injury-table-01-03-2022.pdf. ↩︎
- “Rotarix Package Insert and Patient Information.” GlaxoSmithKline, (2019). https://www.fda.gov/media/75726/download. ↩︎
- Yih, W. Katherine, et al. Ph.D., M.P.H. “Intussusception Risk after Rotavirus Vaccination in U.S. Infants.” New England Journal of Medicine 370, (2014): 503-512. https://www.nejm.org/doi/full/10.1056/NEJMoa1303164. ↩︎
- Yih, W. Katherine, et al. Ph.D., M.P.H. “Intussusception Risk after Rotavirus Vaccination in U.S. Infants.” New England Journal of Medicine 370, (2014): 503-512. https://www.nejm.org/doi/full/10.1056/NEJMoa1303164. ↩︎
- “Rotavirus Vaccine for the Prevention of Rotavirus Gastroenteritis Among Children Recommendations of the Advisory Committee on Immunization Practices (ACIP).” Morbidity and Mortality Weekly Report 48, no. RR-2 (2000): 1-23. https://www.cdc.gov/mmwr/preview/mmwrhtml/00056669.htm. ↩︎
- Wagner, Josef, et al. “Early-life Gut Microbiome Associates with Positive Vaccine Take and Shedding in Neonatal Schedule of the Human Neonatal Rotavirus Vaccine RV3-BB.” Nature Communications 16, (2025). https://www.nature.com/articles/s41467-025-58632-6. ↩︎
- Kling, Jim. “The Vaccine That Almost Wasn’t.” MIT Technology Review, (2005). https://www.technologyreview.com/2005/06/01/230880/the-vaccine-that-almost-wasnt/. ↩︎
- Burnett, Eleanor, et al. “Rotavirus Infection, Illness, and Vaccine Performance in Malnourished Children: A Review of the Literature.” The Pediatric Infectious Disease Journal 40, no. 10 (2021): 930–936. https://stacks.cdc.gov/view/cdc/110382. ↩︎
- “The Impact of Rotavirus Vaccination.” Johns Hopkins University Bloomberg School of Public Health,. https://publichealth.jhu.edu/sites/default/files/2024-02/rota-brief7-vaccineimpact2022ax.pdf. ↩︎
- “Advisory Committee on Immunization Practices February 11-12, 1998 Meeting Verbatim Transcript.” CDC Stacks, (1998). https://stacks.cdc.gov/view/cdc/76564. ↩︎
- Kling, Jim. “The Vaccine That Almost Wasn’t.” MIT Technology Review, (2005). https://www.technologyreview.com/2005/06/01/230880/the-vaccine-that-almost-wasnt/. ↩︎
- Pan American Health Organization. (2006). Report on the XXIII meeting of the Caribbean Expanded Program on Immunization Managers, Jamaica, 2006. PAHO. https://www.paho.org/sites/default/files/Immunization-Caribbean-EPI-23-Mgrs-Mtg-2006-e.pdf ↩︎
- Goodell, Sarah, MA, et al. “Cost Savings and Cost Effectiveness of Clinical Preventive Care.” The Synthesis Project, no. 18 (2009). https://www.walkboston.org/sites/default/files/Cost%20savings%20and%20cost-effectiveness%20of%20clinical%20preventive%20care.pdf. ↩︎
- United States. Advisory Committee on Immunization Practices. (1998, October 21–22). Advisory Committee on Immunization Practices October 21-22, 1998 meeting minutes. (p. 29) ACIP meeting minutes. https://stacks.cdc.gov/view/cdc/76565 ↩︎
- Wagner, Josef, et al. “Early-life Gut Microbiome Associates with Positive Vaccine Take and Shedding in Neonatal Schedule of the Human Neonatal Rotavirus Vaccine RV3-BB.” Nature Communications 16, (2025). https://www.nature.com/articles/s41467-025-58632-6. ↩︎
- U.S. Food and Drug Administration. (2017, November). Regulatory perspective on development of preventive vaccines for global infectious diseases [PDF file]. https://www.fda.gov/media/134489/download ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859. ↩︎
- Murphy, Trudy V. MD., et al. “Intussusception Among Infants Given an Oral Rotavirus Vaccine.” New England Journal of Medicine 344, (2001): 564-572. https://www.nejm.org/doi/10.1056/NEJM200102223440804?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200www.ncbi.nlm.nih.gov. ↩︎
- Schwartz, Jason L. “The First Rotavirus Vaccine and the Politics of Acceptable Risk.” The Milbank Quarterly, (2012) https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-0009.2012.00664.x?msockid=384032df62c169493a03270163b96859.. ↩︎
- Childress, Mark B., et al. “Russell Bruesewitz V. Wyeth, Inc.” The Supreme Court of the United States, (2010). https://www.justice.gov/osg/media/197896/dl?inline. ↩︎
- “FACA: Conflicts of Interest and Vaccine Development — Preserving the Integrity of the Process.” Committee on Government Reform, U.S. House of Representatives, (2000). https://www.govinfo.gov/content/pkg/CHRG-106hhrg73042/html/CHRG-106hhrg73042.htm. ↩︎




